Transport Mechanismus Study of Glassy Electrolytes Using Acoustic Attenuation and Conductivity Spectroscopy
P. Bury 1), M. Jamnický 2), P. Hockicko 1), I. Jamnický 1)
Department of Physics, Žilina University, 010 26 Žilina (bury@fel.utc.sk) 1)
Department of Ceramic,Glass and Cement, Slovak Technical University,812 37 Bratislava 2)
Introduction A particular attention is paid to the experimental study of transport mechanisms in glassy materials with the fast ion transport that play an important role in a number of modern electrochemical devices. The investigation of conductivity spectra of ionic glasses is the basic tool for transport mechanisms study [1]. However, the acoustic attenuation measurement is another very useful technique for non-destructive investigation of transport mechanisms in conductive glasses that can have even some advantages comparing to electrical ones [2,3]. In the contribution we present both acoustic and conductivity spectra of new phosphate glasses containing Cu+ conductive ions in the system CuI-CuBr-Cu2O-P2O5 with respect to investigate ion transport mechanisms and to determine the relation between acoustical and electrical properties considering the various glass compositions.
Experimental The preparation of glasses in the system CuI-CuBr-Cu2O/P2O5 from commercial reagents (Fluka) represented the procedure already described [4]. The system of glasses (Tab. 1) were prepared to investigate the role of cuprous halides produced Cu+ ions keeping their ratio to the glass forming system constant. The acoustical attenuation was measured using MATEC attenuation comparator for longitudinal acoustic wave of frequency 13, 18 and 27 MHz generated by quartz transducer in the temperature range of 140-380 K [5]. The temperature and frequency dependencies of electrical conductivity (dc and ac in the frequency range 50 Hz - 1 MHz were measured using FLUKE PM 6306 impedance analyzer.
Results The results of measured dc conductivity as a function of temperature using
the relation s= s0exp(-Ea /kT), where Ea is the activation energy, k is the Boltzman constant and T is the absolute temperature, indicate at least three transport mechanisms with activation energies Ea1, and Ea2 for higher and Ea3 for lower
Tab. 1. Starting compositions (in mol. %) of prepared glasses and summary of activation energies determinde from both dc conductivity and acoustic spectra.
|
Glass |
|
Composition (in mol.%) |
|
|
|
|
|
|
|
|
|||
|
Sample |
CuI |
CuBr |
Cu2O |
P2O5 |
[eV] |
[eV] |
[eV] |
[eV] |
[eV] |
[eV] |
|||
|
BIDP1 |
15,91 |
2,27 |
54,55 |
27,27 |
0.46 |
0.52 |
0.40 |
0.47 |
0.41 |
0.25 |
|||
|
BIDP2 |
13,63 |
4,55 |
54,55 |
27,27 |
0.44 |
0.54 |
0.44 |
0.46 |
0.41 |
0.27 |
|||
|
BIDP3 |
11,36 |
6,82 |
54,55 |
27,27 |
0.46 |
0.58 |
0.42 |
0.46 |
0.41 |
0.26 |
|||
|
BIDP5 |
9,09 |
9,09 |
54,55 |
27,27 |
0.44 |
0.54 |
0.42 |
0.46 |
0.42 |
0.26 |
|||
|
BIDP6 |
6,82 |
11,36 |
54,55 |
27,27 |
0.39 |
0.52 |
0.43 |
0.46 |
0.42 |
0.28 |
|||
|
BIDP7 |
4,55 |
13,63 |
54,55 |
27,27 |
0.41 |
0.50 |
0.43 |
0.45 |
0.42 |
0.25 |
|||
|
BIDP8 |
2,27 |
15,91 |
54,55 |
27,27 |
0.42 |
0.52 |
0.41 |
0.46 |
0.43 |
0.27 |
|||
|
BDP |
0 |
18,18 |
54,55 |
27,27 |
0.40 |
0.51 |
0.38 |
0.47 |
0.43 |
0.26 |
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
temperatures, respectively. The shapes of experimental Arrhenius plots of dc conductivity indicate also some association and dissociation processes, respectively connected with defects formation [5]. Activation energies calculated from Arrhenius plots of dc conductivity for all glass samples are summarized in Tab. 1. All prepared glasses have high ionic conductivity at room temperatures 10-2-10-4 W-1cm-1.
The both activation energies Ea1 and Ea2 determined at higher temperatures from dc measurements as a function of glass composition for mixed CuI/CuBr halides containing the same mol. % of glass-forming components (Cu2O+P2O5) varies significantly with substitution of cuprous halides. As we can see the investigated glasses exhibit a mixed cation effect manifesting in the activation energies, referred to the reduced long range diffusivities and attributed to interactions between dissimilar ions, to phase separation or to cation sites of different size [6].
The infrared study of Cu+ ions conducting glasses in the same
systems [7] showed that these glasses contain mainly ![]() ,
and tetrahedral anions groups and cuprous halides as well as their mixtures do
not affect significantly the dominant phosphate oxide structural units of
glasses.
,
and tetrahedral anions groups and cuprous halides as well as their mixtures do
not affect significantly the dominant phosphate oxide structural units of
glasses.
The set of frequency dependencies of ac conductivity measured at various temperatures (conductivity spectra) is illustrated in Fig. 1 for glass sample BIDP3 as representative of investigated system. The obtained ac conductivity measurements correspond, because of limited frequency range, only to two regimes (II and III) of complete conductivity spectra due to the hopping motion [1].
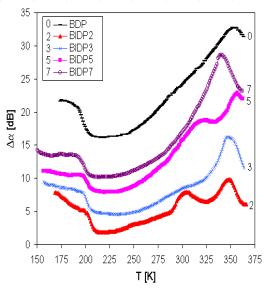
Fig. 1. The conductivity spectra of sample BIDP3.
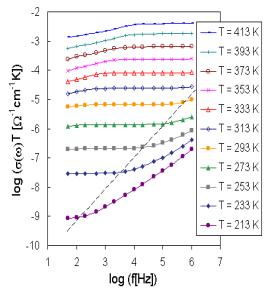
Fig. 2. The set of attenuation spectra measured at frequency 18 MHz.
The glass sample BIDP3 similarly as other samples exhibits one slope of brakes on the ac conductivity spectra indicated another transport hopping process. However, the jump from one value of ac conductivity to another was registered at higher temperatures and lower frequencies, characterizing the transition between two kinds of hopping processes. Low frequency part in regime III and dispersive regime II are due to the hopping motion of the mobile ions and are obviously explained in terms of jump relaxation model considering several kinds of cation sites.
The additional information can be obtained from acoustical spectra that indicate in investigated samples broad attenuation maxima at higher temperatures, in which we can distinguish at least two separated peaks (T1, T2). At higher frequencies (18 and 27 MHz) the peaks are very well recognized and at frequency 27 MHz there were registered even peaks corresponding to transition temperature Tg, that coincide very well with values determined by DTA. One another peak (T3) was registered also at lower temperatures. The change of position and proportion of the peaks maxima can be seen in Fig. 2 illustrating the acoustic attenuation spectra measured at 18 MHz. The attenuation spectra can be explained by the assumption that temperature peaks are caused by the relaxation processes of mobile Cu+ ions in connection with different kinds of sites.
The peaks at the broad maxima of acoustical attenuation spectra indicate at
least two basic possible transport mechanisms with the activation energies that
are shifted comparing to those determined in d.c. electric
measurement. All glasses we studied using acoustic
spectroscopy exhibit an Arrhenius–type relaxation between the peak temperature Tpeak
and the applied frequency ![]() , where
, where ![]() is the activation energy, k is
Boltzmann constant and n0
is the pre-exponential factor. Using the values of Tpeak for
individual peaks, the activation energies
is the activation energy, k is
Boltzmann constant and n0
is the pre-exponential factor. Using the values of Tpeak for
individual peaks, the activation energies ![]() were
determined (Tab. 1). However, it seems, that individual transport
mechanisms indicated by acoustical spectra does not correspond to the
mechanisms indicated by dc measurement.
were
determined (Tab. 1). However, it seems, that individual transport
mechanisms indicated by acoustical spectra does not correspond to the
mechanisms indicated by dc measurement.
Conclusions The experimental investigation of ion conductive glasses in the system CuI-CuBr-Cu2O-P2O5 proved that acoustical spectroscopy in connection with conductivity spectroscopy are very useful techniques for transport and relaxation mechanisms study in these materials. Several different kinds of sites responsible for ionic hopping motion were discovered and the influence of chemical composition on ion transport mechanisms was exhibited. The activation energies of some transport mechanisms were determined, too. Further theoretical analysis of experimental measurements using suitable relaxation model should be done for better understanding of ion transport mechanisms in this type of conductive glasses.
References
[1] K. Funke, B. Roling, M. Lange, Sol. State Ionics 105 (1998) 195.
[2] M. Cutroni and A. Mandanici, Sol. St. Ionics, 105 (1998) 149.
[3] P. Bury, P. Hockicko, M. Jamnický and I. Jamnický, Advances in Electrical and Electronics Engineering, Vol. 2, No 3-4 (2003) 16.
[4] P. Znasik, M. Jamnicky, J. Mat Science Letter 14 (1995) 766.
[5] P. Bury, P. Hockicko, M. Jamnický and I. Jamnický, Proc. 18th Int. Congress on Acoustics, KYOTO 2004 (will be published).
[6] B. Roling, A. Happe, M. D. Ingram and K. Funke, J. Phys. Chem. B 103 (1999) 4122.
[7] P. Znasik, M. Jamnicky, Solid St. Ionics, 92 (1996) 145.